Modeling the Spinal Cord as a Peripheral Nerve Suggests to Repair Complete Transection Injuries by an Artificial Device
Antonio Merolli, MD FBSE1, Duke Shereen, PhD2 and Joachim Kohn, PhD FBSE1, (1)Rutgers - The State University of New Jersey, Piscataway, NJ, (2)City University of New York, New York, NY
Introduction: Artificial nerve guides have been used in the shape of rigid polymeric containers; they were able to repair complete transections injuries of single peripheral nerves and of whole brachial plexus. The concept is that the implantable device provides a suitable biophysical environment that deceives against its outside the massive scar tissue reaction. In this way, the device protects the axonal regeneration process that occurs inside. Can a similar approach work in complete transection injuries of the spinal cord? This hypothesis has been tested in the spinal cord of the rabbit, whose diameter (about 2.5 mm) falls in the range of artificial nerve guides (conduits) commonly used in hand surgery.
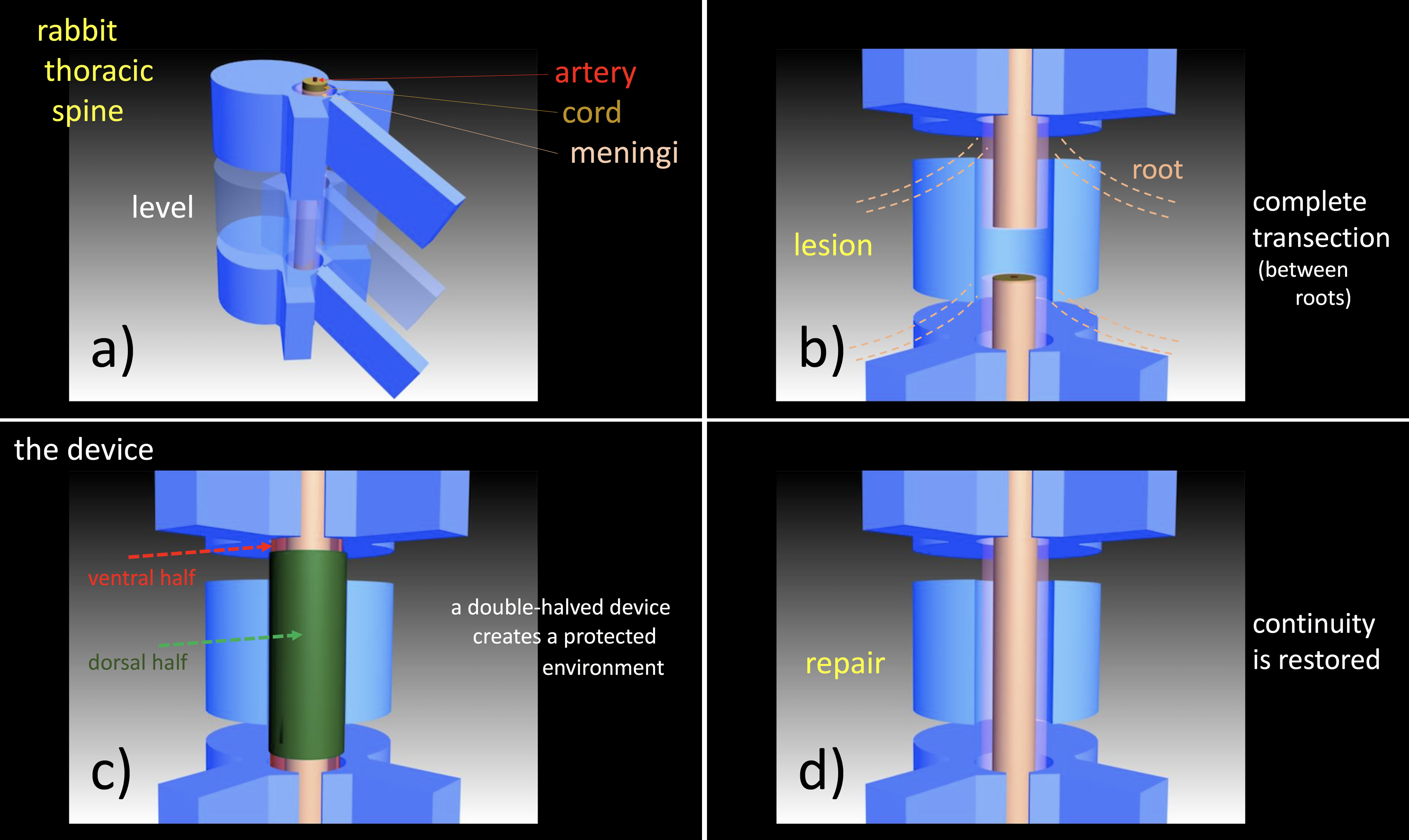
Materials and Methods: A cylindrical shaped 3D printed degradable device made of a Tyrosine-derived polycarbonate was implanted into a New Zealand White vertebral canal at the level of the lower thoracic vertebra T9 (figure 1). A complete transection injury was produced after acces to the cord was gained by a careful laminectomy performed by a piezoelectric saw. Magnetic Resonance Imaging and Tractography were performed by a 9.4 Tesla equipment. Myelin Basic Protein, Choline acetyltransferarse, Glial Fibrillary Acidic Protein were immunostained on wax-embedded EDTA-decalcified section of the whole spine. Untreated lesions were compared as control.
Results: The 3D printed degradable device can be implanted in the New Zealand White rabbit spinal canal in a way that creates a defined protective environment. Preliminary results show how injuries treated by the artificial device developd minimal fibroblastic and astroglial scars in comparison with controls. Immunohistochemical slides and MRI analysis showed a high degree of correlation.
Conclusions: While there are differences in cellularity and metabolic pathways between peripheral nerves and spinal cord, there is now an amount of references that shows how axons in spinal cord injuries can regenerate and bridge the gap in favourable circumstances. This model will help in the study of fundamental mechanisms in spinal cord regeneration and repair. Moreover, this model will test a new surgical approach aimed at being translated into clinical practice. This device-assisted procedure can integrate and potentiate current therapeutic approaches and it is not proposed as an alternative to them.
Figure 1. In: a)-T9 is the level of the lesion; b)-a complete transection injury is produced in the length between the roots; c)-an artificial nerve guide is put in place; d)-after the regeneration process the device gradually degrades and disappears.
Back to 2020 ePosters